Previously, we introduced coating selection for NdFeB magnets and covered surface treatments such as electroplating and phosphating. Here, we focus on electrophoretic coating.
Electrophoretic coating is a widely used anti-corrosion surface treatment for both sintered and bonded NdFeB magnets. The parts are immersed in a water-based coating bath containing electrodes. When DC current is applied, an electrochemical reaction causes the coating to deposit uniformly on the surface, forming a polymer-based anti-corrosion layer.
This coating offers strong adhesion to porous magnet surfaces and provides excellent resistance to salt spray, acids, and alkalis. However, its resistance to high temperature and humidity is relatively weaker.
1.High material utilization:
Compared to spray coating (where ~50% of paint may be lost), electrophoresis can achieve 90–95% utilization through ultrafiltration.
2.Environmentally friendly:
Water-based coating eliminates the need for organic solvents, reducing pollution and material consumption.
3.High efficiency:
After pre-treatment, bonded magnets can be coated without drying, shortening processing time.
4.Uniform coating on complex shapes:
Electric field-driven deposition ensures consistent coating even on intricate geometries.
5.Good coating quality:
Strong adhesion and excellent corrosion resistance.
1.High initial investment and complex control:
Bath maintenance requires strict control of process parameters.
2.Surface defects not masked:
Cannot cover defects such as pores, scratches, or dents—high surface quality is required.
3.Labor-intensive handling:
Hanging and unloading parts require manual work.
Types of Electrophoretic Coating
Electrophoretic coatings consist of polymer resins, solvents, pigments, and water, forming a uniform suspension.
There are two main types:
• Anodic E-coating:
The magnet acts as the anode. During coating, both the base metal and surface layer may dissolve, reducing corrosion resistance. Therefore, it is less commonly used.
• Cathodic E-coating (most common):
The magnet acts as the cathode, using cationic coatings formed by neutralizing amino-functional resins with organic acids.
oLower toxicity
oBetter mechanical strength and adhesion than anodic coatings
oHigher cost
oRequires corrosion-resistant equipment (e.g., stainless steel or acid-resistant plastics) due to acidic bath conditions
Process Flow of Electrophoretic Coating for Sintered NdFeB
The electrophoretic coating process for sintered NdFeB magnets is shown below and mainly consists of three stages: surface pre-treatment, electrophoretic deposition, and drying & curing.
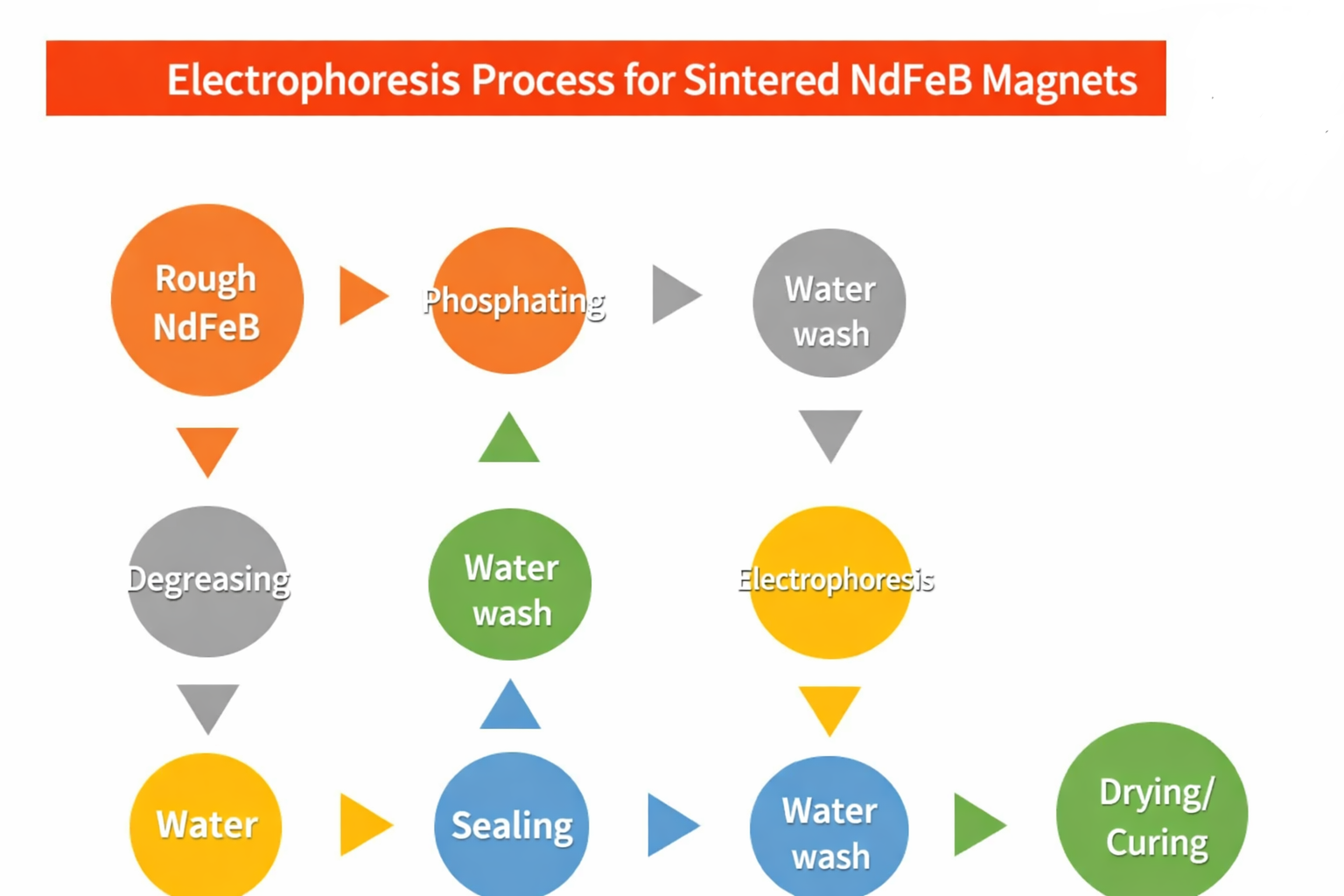
A smooth and clean surface is the foundation for high-quality electrophoretic coating. The purpose of surface treatment is to create optimal surface conditions.
Electrophoresis is a complex chemical process. As the coating continuously deposits on the magnet surface, the composition of the bath also changes. Therefore, parameters such as solid content, bath pH, temperature, voltage, and electrode spacing must be strictly controlled.
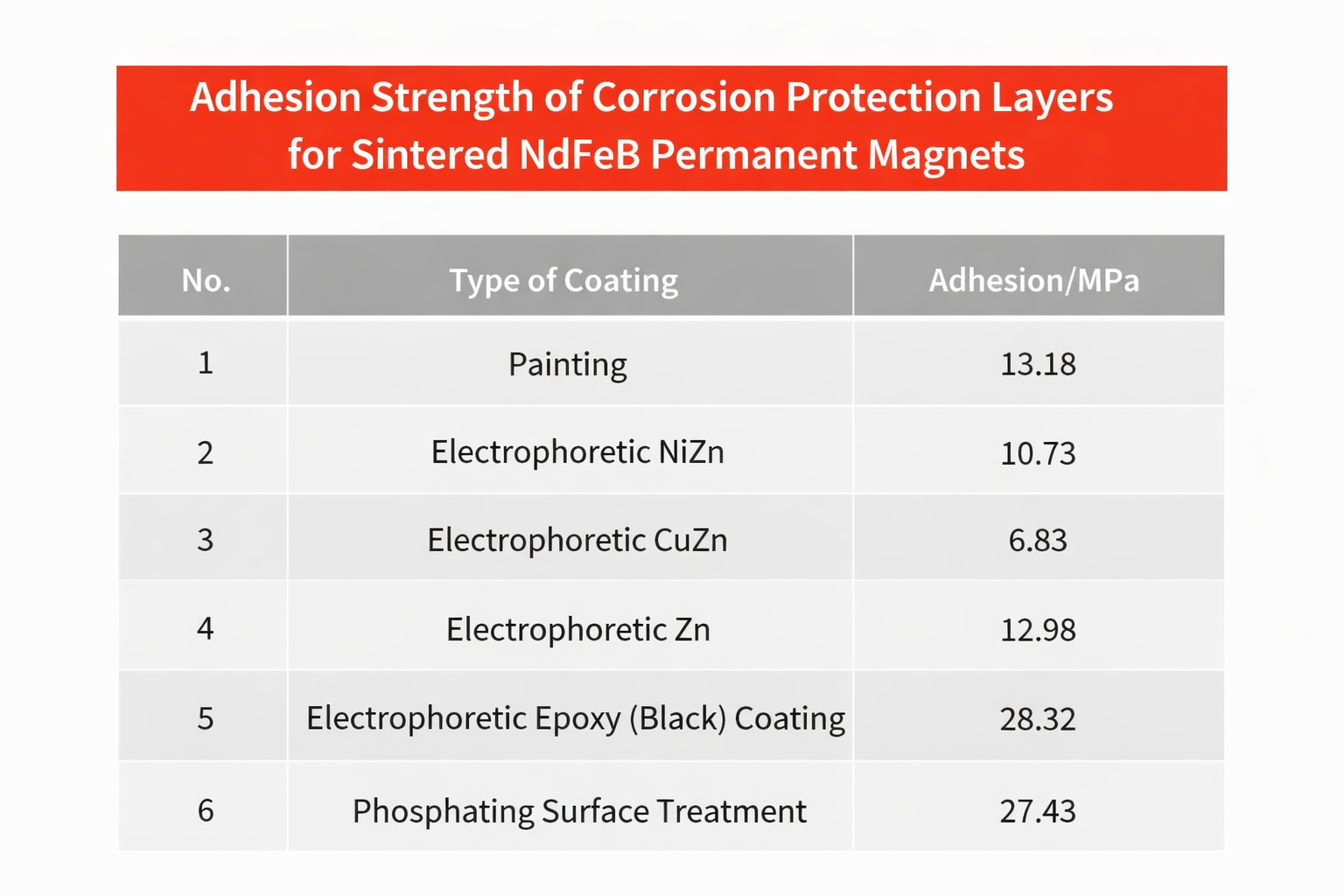
Comparison of Adhesion between Different Coatings and the Magne