The generation of magnetic fields can be broadly categorized into two main aspects: the first is based on moving electric currents (electromagnetic induction), while the second is based on the intrinsic spin of the fundamental particles that constitute matter. The first category—the magnetic effect of electric currents—is relatively familiar to us; when an electric current flows through a conductor, the directional movement of free electrons generates a magnetic field. The second category—magnetic fields generated by the matter itself—is the primary subject of our discussion today.
Everything in the universe possesses magnetic properties—from the small objects around us, such as tables and chairs, to the vast celestial bodies of the cosmos, such as planets and the Sun; without exception, all exhibit magnetism. Regardless of their physical state (crystalline, amorphous, liquid, or gaseous), or the conditions under which they exist (high or low temperature, high or low pressure), they all possess magnetic properties. The only difference lies in the strength of this magnetism: some substances exhibit strong magnetism, while others exhibit weak magnetism; however, it can be stated that no substance exists that is entirely devoid of magnetic properties.
Based on the characteristics they exhibit when placed within an external magnetic field, substances can be classified into five categories: paramagnetic, diamagnetic, ferromagnetic, ferrimagnetic, and antiferromagnetic substances. What, then, is the underlying reason why all matter possesses magnetic properties? And what accounts for the distinct characteristics exhibited by these different substances? To answer this, we must begin with the fundamental building blocks of matter: atoms.
Matter is composed of atoms, and atoms, in turn, consist of an atomic nucleus and electrons. Within an atom, electrons possess an *orbital magnetic moment* by virtue of their motion around the nucleus, and a *spin magnetic moment* by virtue of their intrinsic spin. The net magnetic moment of an atom is derived primarily from the sum of these electron orbital and spin magnetic moments; this constitutes the fundamental source of magnetism in all matter. (It is worth noting that the magnetic moment of the atomic nucleus is merely 1/1,836.5 times that of the electron's magnetic moment; consequently, the contribution of the nuclear magnetic moment is generally considered negligible.)
**Magnetic Moments of Single, Isolated Atoms**
A magnetic moment is a directional vector quantity. The electron spins within an atom are classified into two types: spin-up and spin-down. In most substances, the number of electrons with spin-up is equal to the number with spin-down; consequently, the magnetic moments generated by these electrons cancel each other out, resulting in the entire atom exhibiting no net external magnetism. Only in a few substances do the internal electrons possess unequal numbers of spin-up and spin-down electrons. In such cases, after the magnetic moments of the oppositely spinning electrons cancel each other out, a residual portion of the spin magnetic moments remains uncancelled, endowing the entire atom with a net magnetic moment. The magnetic moment of a single atom is determined by its atomic structure—specifically, the arrangement and number of its electrons; indeed, the atoms of all elements in the periodic table possess their own intrinsic magnetic moments.
**Magnetic Moments of Atoms in Crystals**
The preceding discussion focused on the magnetic moments of single, isolated atoms. However, in solid crystalline or amorphous materials, atoms occupy specific lattice sites. These atoms are subject to the influence of the nuclear electric fields and electron electrostatic fields of their neighboring atoms; consequently, the magnetic moments of atoms within a crystal differ from those of single, isolated atoms. Consider, for example, iron, cobalt, and nickel—collectively known as 3d transition metals. Within their crystalline structures, the electrons of certain atoms become delocalized, acting as shared electrons among adjacent atoms. This alteration in the atomic electronic structure results in the "freezing" of a portion of the orbital magnetic moments. Consequently, the magnetic moment of an atom within the crystal is contributed solely by its spin magnetic moment, leading to a divergence between the observed magnetic moment of the atom in the crystal and its theoretical value.
**Magnetic Moments of Macroscopic Materials**
Based on the preceding content, we now understand that everything in the universe possesses magnetic properties, and that magnetism fundamentally originates from atomic-level magnetism. Because different atoms possess distinct magnetic moments, interactions occur between the atomic magnetic moments within macroscopic materials, leading to varying arrangements of these moments at room temperature. Based on their macroscopic magnetic characteristics, we classify materials into five categories: paramagnetic, diamagnetic, ferromagnetic, ferrimagnetic, and antiferromagnetic substances. These characteristics are defined by the following three parameters:
1. Magnetization (M)
The macroscopic magnetic properties of a material are derived from the collective contributions of the magnetic moments of its constituent atoms or molecules. We define the total magnetic moment per unit volume of a material as its *magnetization*, denoted by the symbol *M*, with units of A/m. Let us assume a certain substance has a volume *V* and contains *n* atoms, where each atom possesses a magnetic moment of *μJ*. The total magnetic moment is then given by *M* = μJ1 + μJ2 + ... + μJn—that is, *M* = ΣμJ/ V.
2. The Magnetization Curve (M–H Curve)
When the external magnetic field is zero, the atomic magnetic moments may be arranged in a disordered fashion. However, when a non-zero external magnetic field is applied, the influence of this field causes each atomic magnetic moment to align itself in the direction of the external field; consequently, the magnetization *M* of the substance undergoes a change. The curve illustrating the relationship between the magnetization *M* and the varying external magnetic field *H* is known as the magnetization curve, abbreviated as the M–H magnetization curve. The magnetization curves for different substances vary depending on the material.
3. Magnetic Susceptibility χ
On the M–H magnetization curve, the ratio of M to H at any given point is termed magnetic susceptibility, denoted by χ. Mathematically, χ = M/H; since both M (magnetization) and H (magnetic field strength) are measured in units of A/m, χ represents a relative magnetic susceptibility and is therefore a dimensionless quantity.
We utilize parameters such as the magnitude and alignment of atomic magnetic moments, the shape of the M–H magnetization curve, and magnetic susceptibility to characterize the magnetic properties of substances and to classify them accordingly.
Paramagnetic Substances
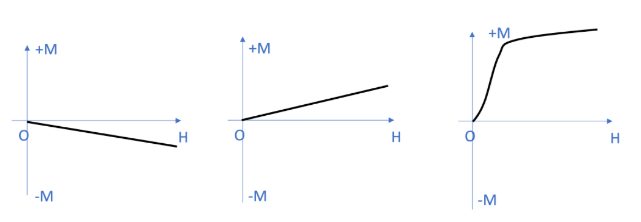
These are substances that, when brought into the vicinity of a magnetic field, undergo magnetization aligned with the direction of the field; however, this magnetization is extremely weak—so faint, in fact, that it can only be detected using precision instruments. Furthermore, if the external magnetic field is removed, the internal magnetization of the substance immediately reverts to zero, resulting in a complete loss of magnetic properties. Examples include aluminum and oxygen gas.
In paramagnetic substances, every individual atom possesses a magnetic moment; consequently, these substances are characterized by inherent atomic magnetic moments. However, there are no significant interactions between adjacent atoms in paramagnetic materials. As a result, at room temperature, the atomic magnetic moments are randomly oriented; the net projection of the atomic magnetic moments (μJ) in any given direction is therefore zero. When an external magnetic field H is applied, the atomic magnetic moments within these substances rotate—albeit by a very small angle—to align with the direction of the external field. Consequently, the magnetization intensity of the material increases gradually as the strength of the external magnetic field increases. The magnetic susceptibility of paramagnetic substances is positive (χ > 0), with typical values ranging from 10⁻⁵ to 10⁻³.
Rough estimates suggest that, in order to achieve complete alignment of the atomic magnetic moments in a paramagnetic substance with the direction of an external field, a magnetic field strength of approximately 10⁹ to 10¹⁰ A/m would be required—a magnitude that remains exceedingly difficult to achieve using currently available artificial magnetic field technologies.
Diamagnetic Substances
These are substances characterized by a negative magnetic susceptibility; in other words, when subjected to a magnetic field, the resulting induced magnetization is directed *opposite* to the direction of the external magnetic field. All organic compounds exhibit diamagnetism; examples of diamagnetic substances include graphite, lead, and water. For diamagnetic substances, the projections of both atomic orbital magnetic moments and spin magnetic moments within a magnetic field are zero; in other words, diamagnetic substances possess no net atomic magnetic moment. However, under the influence of an external magnetic field, the electron orbits generate an induced additional magnetic moment. This induced moment is directed opposite to the external magnetic field, resulting in the manifestation of negative magnetism. The direction of magnetization in diamagnetic substances is negative—opposing the external magnetic field—and its absolute magnitude increases linearly as the strength of the external magnetic field increases.
Ferromagnetic Substances
Ferromagnetic substances are materials that, once magnetized by an external magnetic field, retain their magnetized state—and thus their magnetic properties—even after the external field is removed. To date, 83 metallic elements have been identified; among these, four elements exhibit ferromagnetism at temperatures above room temperature: iron, cobalt, nickel, and gadolinium. Additionally, at extremely low temperatures, five other elements can transition into a ferromagnetic state: terbium, dysprosium, holmium, erbium, and thulium.
In ferromagnetic substances, atoms possess intrinsic atomic magnetic moments. Some electrons become delocalized (shared), causing the spin magnetic moments of adjacent atoms to align parallel to one another in the same direction—a phenomenon known as spontaneous magnetization. The M-H magnetization curve for ferromagnetic substances is nonlinear, and their magnetic susceptibility (χ) varies in response to changes in the magnetic field. Ferromagnetic substances exhibit very high magnetic susceptibility, with values potentially reaching magnitudes of 10⁵ to 10⁷.
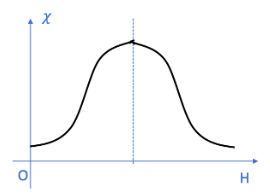
Antiferromagnetic Materials
Antiferromagnetic materials do not generate a magnetic field. Such substances are relatively uncommon, and new antiferromagnetic materials are continuously being discovered. Most antiferromagnetic materials exist only under low-temperature conditions; if the temperature exceeds a certain threshold, they typically transition into a paramagnetic state. For instance, elements such as chromium and manganese exhibit antiferromagnetic properties.
In antiferromagnetic materials, atoms also possess intrinsic atomic magnetic moments. While some electrons become delocalized, the magnetic moments of adjacent atoms align in opposite directions (a phenomenon known as antiferromagnetic ordering). The M–H magnetization curve for antiferromagnetic materials is linear, and their magnetic susceptibility (χ) is positive. The value of χ is typically very small—approximately 10⁻⁴ to 10⁻⁵—and remains constant. This implies that when an antiferromagnetic material is subjected to an external magnetic field, the response of its atomic magnetic moments to the field is minimal; in this regard—and like paramagnetic materials—they are classified as weakly magnetic substances. Furthermore, the magnetic susceptibility (χ) of antiferromagnetic materials varies with temperature, as illustrated in the figure below; the temperature denoted as Tₙis referred to as the Néel temperature.
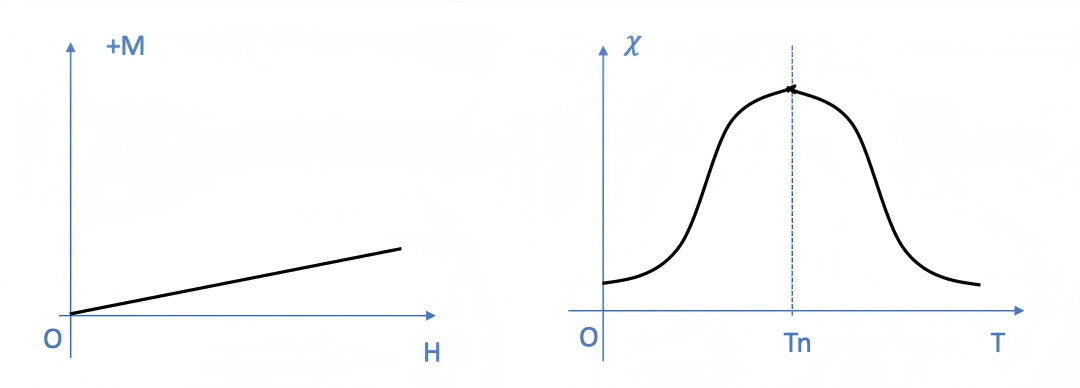
Ferrimagnetic Materials
Their macroscopic magnetic properties are identical to those of ferromagnetic materials, differing only in their slightly lower magnetic susceptibility (with a susceptibility value, χ, ranging from 10² to 10⁵). Typical examples of ferrimagnetic materials include ferrites; the most significant distinction between them and ferromagnetic materials lies in the differences in their internal magnetic structures (specifically, the arrangement of magnetic moments).
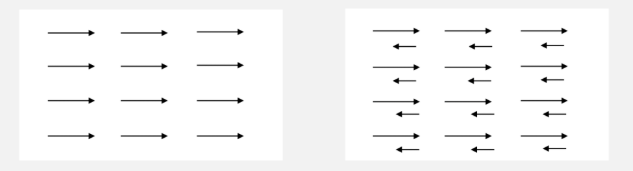
In ferrimagnetic materials, the atomic magnetic moments are non-zero; indirect exchange interactions—or RKKY exchange interactions—exist between adjacent atomic moments, causing the moments of neighboring sublattices to align antiparallel to one another. However, the magnitudes of the atomic moments in these adjacent sublattices differ (as illustrated in the figure). This phenomenon is referred to as ferrimagnetic ordering or spontaneous ferrimagnetic magnetization. The M-H magnetization curves of ferrimagnetic materials are nonlinear—similar to those of ferromagnetic materials—though their magnetic susceptibility is slightly lower; nevertheless, they are still classified as strongly magnetic materials.