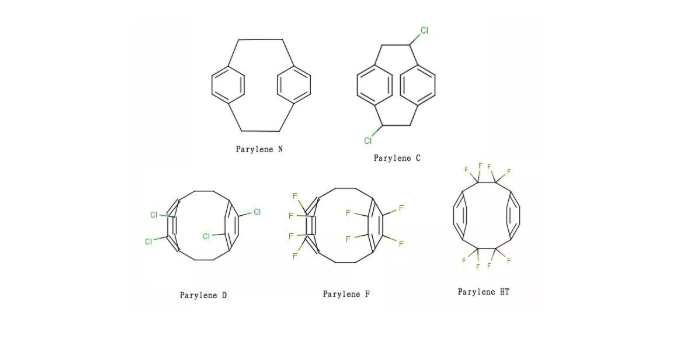
Parylene is a conformal coating material developed and applied by Union Carbide in the mid-1960s. It is a polymer of para-xylene. Based on molecular structure, Parylene is classified into several types, including N, C, D, F, and HT.
Parylene is a protective polymer material, known in Chinese as poly-para-xylylene. It is deposited through vapor-phase deposition under vacuum conditions.
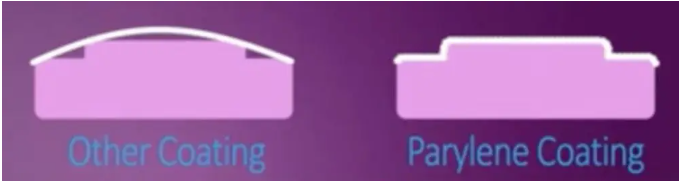
Due to the excellent penetration capability of its active molecules, Parylene can form a pinhole-free, uniform, transparent insulating coating on all surfaces of a component—including internal, recessed, and surrounding areas. This provides comprehensive protection against acids, alkalis, salt spray, fungi, and various corrosive gases.
Since Parylene is not a liquid, it does not exhibit pooling, bridging, or meniscus effects during the coating process.
NdFeB permanent magnets are high-performance materials with strong magnetic properties, but they are also chemically unstable in air. For larger magnets, electroplating or epoxy electrophoretic coatings are commonly used for protection. However, for small or ultra-small magnets—especially ring-shaped and tubular types—these traditional methods often fail to meet performance requirements.
Thanks to its unique deposition process and excellent properties, Parylene can provide complete, conformal coating without weak points, even on very small and complex-shaped magnets. Magnets coated with Parylene can withstand immersion in hydrochloric acid for over 10 days without corrosion. Therefore, Parylene is widely used internationally as an insulating and protective coating for small and ultra-small magnetic components.

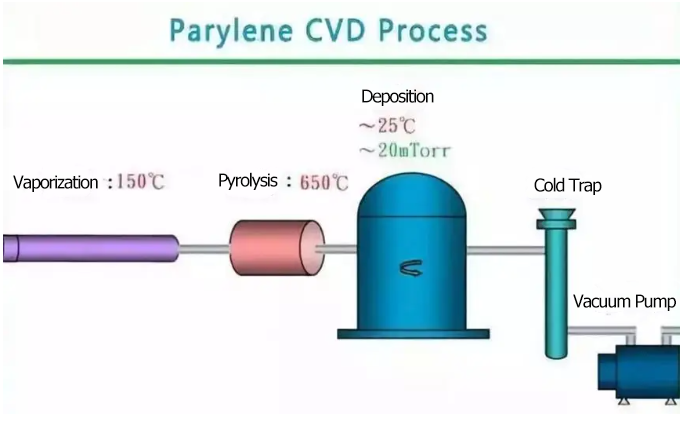
Parylene coatings are typically applied using Chemical Vapor Deposition (CVD), a process where gaseous reactants undergo chemical reactions and deposit as a solid film directly onto the substrate surface.
The process consists of three main steps: